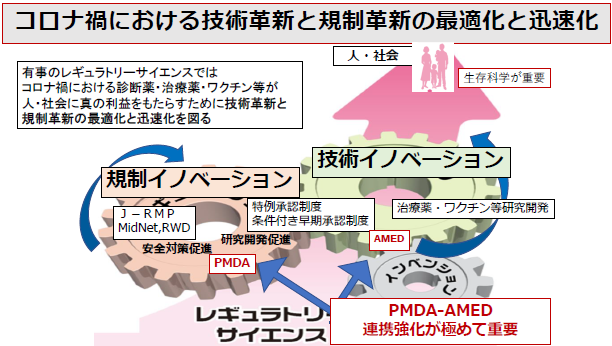
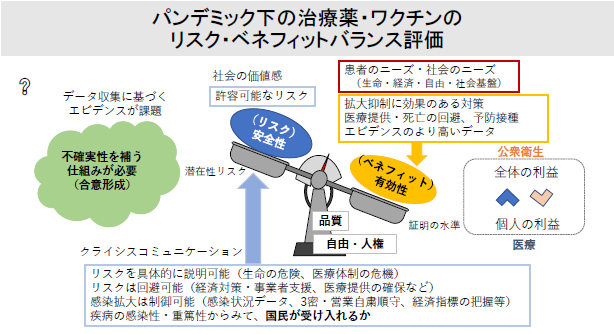
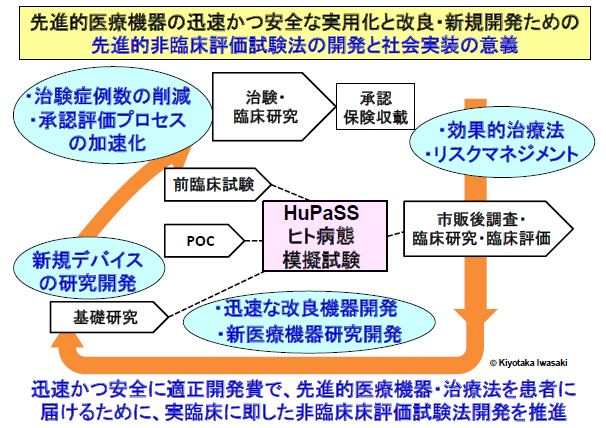
Research Theme
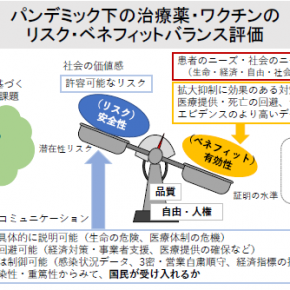
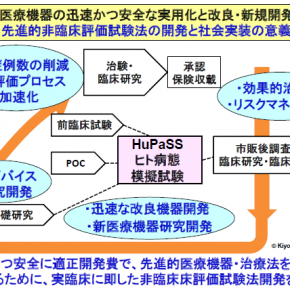
The Institute for Medical Regulatory Science promotes research on the regulation of medical devices, pharmaceuticals, and regenerative medicine products, as well as on non-clinical testing, clinical trials, evaluation methods, and the appropriate use of therapeutic medical devices in real-world clinical settings. In particular, we are advancing research and development on experimental systems and evaluation methodologies that simulate human pathological conditions to accelerate the development of Class IV therapeutic medical devices. Additionally, in collaboration with academia and startups, we are engaged in regulatory science initiatives that facilitate the societal implementation of Class IV therapeutic medical devices, playing a key role in fostering an ecosystem that transitions basic research into practical applications. Our efforts focus on the global development of innovative Class IV therapeutic medical devices, such as tissue regenerative devices based on decellularized tissue, which promote in vivo autologous tissue construction. Together with researchers from diverse fields and invited experts, we are also addressing the regulation of “Software as a Medical Device” for medical and health applications, driving innovation through regulatory science research.
Research Director
IWASAKI, Kiyotaka
Faculty of Science and Engineering, Graduate School of Advanced Science and Engineering
Project Members
- KASANUKI, Hiroshi
- UMEZU, Mitsuo
- HIGASHIDA, Masayuki Research Associate, Comprehensive Research Organization,
- SHIOZAKI, Yuji Research Associate, Comprehensive Research Organization,
- AYABE, Hironori Professor, School of Creative Science and Engineering
- HASUIKE, Takashi Professor, School of Creative Science and Engineering
- HATTORI, Kaoru Associate Professor(non-tenure-track), Graduate School of Advanced Science and Engineering
- HIGASHIDE, Hironori Professor, Graduate School of Business and Finance
- IMAI, Shinya Research Associate, Graduate School of Advanced Science and Engineering
- ISHII, Hiroyuki Professor, School of Creative Science and Engineering
- IWASAKI, Kiyotaka Professor, Graduate School of Advanced Science and Engineering
- KOMATSUBARA, Akinori Professor, School of Creative Science and Engineering
- NOGUCHI, Haruko Professor, School of Political Science and Economics
- TAKANISHI, Atsuo Professor, School of Creative Science and Engineering
- TAKEOKA, Shinji Professor, School of Advanced Science and Engineering
- YAMAGUCHI, Nariaki Professor, School of Law
- YOKONO, Megumu Associate Professor, School of Social Sciences
- DEGUCHI, Atsuko
- ECHIGO, Masahiro
- FUKAMIZU, Junichi
- HANAMURA, Nobuyuki
- HASHIMOTO, Yoshihide
- HIKICHI, Yutaka
- HIRANO, Asao
- IIJIMA, Kazuo
- IKENO, Fumiaki
- INOUE, Tomoko
- ITO, Masafumi
- ITOU, Junya
- IWAISHI, Chie
- JOKURA, Yoji
- KATAYAMA, Ikuo
- KATO, Reiko
- KATO, Tsugiko
- KAWAKAMI, Akihiko
- KAWAMOTO, Yukiyoshi
- KEARON, Emi Matsuoka
- KOJIMA, Shigehiro
- KOKUBO, Akira
- KOMINE, Teruo
- KONDO, Hideyuki
- KUNO, Takatoshi
- MASAMUNE, Ken
- MATSUMOTO, Keisuke
- MATSUMOTO, Toru
- MATSUMOTO, Toru
- MATSURA, Yuka
- MIYAJIMA, Atsuko
- MORI, Mitsuko
- MURAGAKI, Yoshihiro
- MURASE, Gen
- NAKAO, Koji
- NAKAOKA, Ryusuke
- NAKAYAMA, Masafumi
- NOMURA, Kenichi
- NOMURA, Yusuke
- OKAMURA, Takayuki
- ONO, Mao
- ONOMURA, Akira
- ORIBE, Yusuke
- OTAKE, Masanori
- OTSU, Hiroshi
- OTSU, Ryoji
- SAKODA, Hideyuki
- SASE, Kazuhiro
- SOGABE, Kazumi
- SORIMACHI, Kazuko
- SUZUKI, Hiroyuki
- TAKATORI, Fumihiko
- TAMURA, Manabu
- TANAKA, Yutaka
- TANIGUCHI, Yasuhiro
- TSUBOKO, Yusuke
- TSUYUKI, Kenichiro
- UEMATSU, Miyuki
- UEMURA, Nobuo
- YAHAGI, Kazuyuki
- YAJIMA, Toshitaka
- YAMADA, Naoki
- YAMAMOTO, Eiichi
- YAMAMOTO, Tadashi
- YAMASHITA, Shingo
- YAMATO, Masayuki
- YAMAWAKI, Masahiro
- YUBA, Mitsuru
Research Keywords
Regulatory Science, Evaluation Science, Human Pathological Simulator and System, Medical Innovation, Social Implementation
Research Summary
The Institute for Medical Regulatory Science promotes research on the regulation of medical devices, pharmaceuticals, and regenerative medicine products, as well as non-clinical testing, clinical trials, evaluation methods, and the appropriate use of therapeutic medical devices in real-world clinical settings.
In particular, we advance research and development on experimental systems and evaluation methodologies that simulate human pathological conditions. These systems are applied to:
(1) The development of First-in-Class Class IV therapeutic medical devices
(2) The assessment of efficacy and safety in regulatory approval applications
(3) The safe and effective post-approval use of novel medical devices
Through the development and application of human pathological simulator and systems, we play a pivotal role in fostering an ecosystem that promotes both the research and development of innovative therapeutic medical devices and their societal implementation.
Furthermore, we accelerate the research and development of Class IV therapeutic medical devices, including biological decellularized tissues that are replaced by autologous tissues in vivo, driving regulatory science research to support their integration into society.
We also conduct research on the regulation of “Software as a Medical Device” (SaMD), which is expected to improve health and medical care outcomes.
Our institute includes researchers from diverse fields such as engineering, medicine, pharmacy, law, economics, ethics, business, and social sciences, along with invited researchers from academia, clinical hospitals, medical-related companies, national research institutes, government agencies, and startups.
Together, we engage in regulatory science research aimed at shaping policies for medical technologies that will contribute to a better global future.