Oral bacteria in the gut could drive immune cell induction and inflammatory bowel disease
Tue, Nov 14, 2017-
Tags
On average, we humans ingest 1.5 liters of saliva containing bacteria every day. Could this possibly have harmful effects on our health?
According to Professor Masahira Hattori of Waseda University, the answer is yes. “We noticed that oral microbes are relatively enriched in gut microbiomes of patients with several diseases such as inflammatory bowel disease (IBD), HIV infection, and colon cancer compared with healthy individuals.” However, the causal role of oral bacteria in the intestine has remained unclear.
Professor Hattori and Professor Kenya Honda of Keio University School of Medicine led research to investigate the relationship between salivary microbes and IBD, and they found that when the bacterium Klebsiella pneumoniae colonizes in an intestine out of microbial balance, immune cells called T helper 1 (TH1) become overactive in the gut, resulting in intestinal inflammation leading to the onset of IBD. Their findings suggest a profound association between oral microbe and gut ecosystem, which provides new insights into microbiome research, and advance development of a novel type of medicine in therapeutics of chronic inflammatory diseases.
This study was published in the online version of Science on October 20.
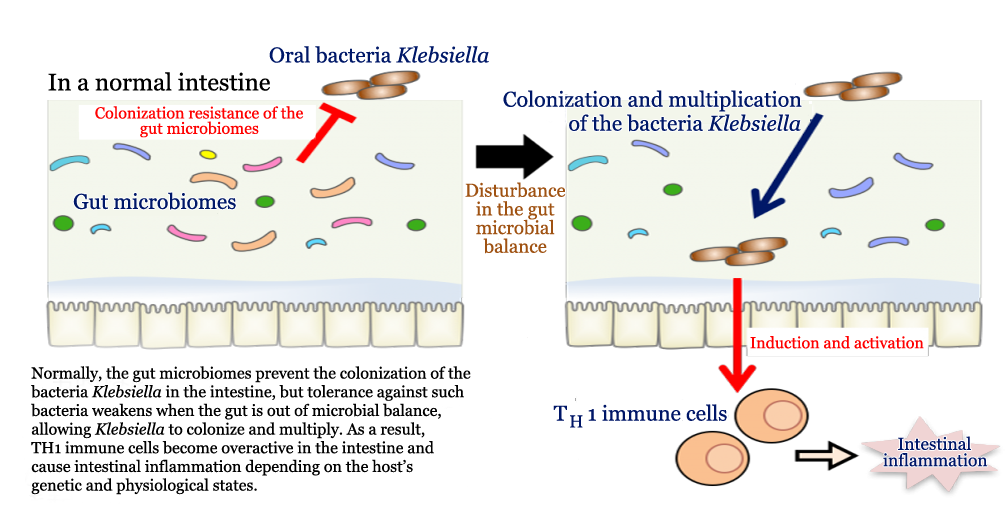
Specifically, they administered salivary samples of healthy and IBD-afflicted individuals to germ-free mice respectively. After six to eight weeks, the microbial content of the mice’s feces were analyzed by 16S ribosomal RNA gene sequencing, which showed that approximately 20 kinds of oral bacteria were present in these mice. Furthermore, an immunological analysis found that the level of TH1 cells, characteristically expressing interferon-gamma protein which activates microphage to enhance bactericidal activities but could be an onset of autoimmune diseases when excessive, significantly increased in some of the mice.
To survey exactly which bacteria induced TH1 cells in the gut of the TH1 induced mice the researchers isolated and cultured the microbes from the feces, and then separately inoculated each microbe to germ-free mice. Through this experiment, they found that Klebsiella pneumoniae was a strong inducer of TH1 cells when colonizing in the gut.
Klebsiella pneumoniae is usually a normal resident in the mouth of healthy individuals, but as demonstrated in an experiment on specific pathogen-free mice treated and untreated with the antibiotic ampicillin, they can colonize in the gut and activate TH1 cells when antibiotics disturb the gut microbial balance and weaken tolerance for the colonization of oral bacteria reaching the intestine. “It is advisable to avoid excessive, long-term use of antibiotics for this reason, even for healthy people,” Professor Hattori points out. Moreover, further experiments showed that the colonization of Klebsiella pneumoniae in the gut induces TH1 cells, which further elicits inflammation depending on the host’s genetic and physiological states. This is because, on treating with the antibiotic ampicillin, no inflammation was observed in wild-type SPF mice with the induction of TH1 cells as described above, but it was caused in IL (interleukin)-10-deficient SPF mice. IL-10 acts to suppress the inflammation.
Some may think having good oral hygiene to eliminate Klebsiella pneumoniae will help them stay healthy, but unfortunately, Klebsiella pneumoniae is multi-drug resistant, and ways to specifically kill or remove it do not presently exist. However, Professor Hattori says, “Although more research needs to be done, our results will help the development of very narrow spectrum antibiotics, lytic bacteriophages specifically targeting Klebsiella pneumoniae, and a way to improve the weakened colonization resistance of the gut microbiomes, for patients with chronic inflammatory diseases including IBD.”
Reference
- Published in the online version of Science on October 20, 2017
- Title: Ectopic colonization of oral bacteria in the intestine drives TH1 cell induction and inflammation
- Authors: Koji Atarashi (1,2), Wataru Suda (1,3,4), Chengwei Luo (5,6), Takaaki Kawaguchi (1,2), Iori Motoo (2), Seiko Narushima (2), Yuya Kiguchi (3), Keiko Yasuma (1), Eiichiro Watanabe (2), Takeshi Tanoue (1,2), Christoph A. Thaiss (7), Mayuko Sato (8), Kiminori Toyooka (8), Heba S. Said (4,9), Hirokazu Yamagami (10), Scott A. Rice (11), Dirk Gevers (5), Ryan C. Johnson (12), Julia A. Segre (12), Kong Chen (13), Jay K. Kolls (13), Eran Elinav (7), Hidetoshi Morita (14), Ramnik J. Xavier (5,6), Masahira Hattori* (3,4), Kenya Honda* (1,2)
1) Department of Microbiology and Immunology, Keio University School of Medicine, 35 Shinanomachi, Shinjuku-ku, Tokyo 160-8582, Japan
2) RIKEN Center for Integrative Medical Sciences, 1-7-22 Suehiro-cho, Tsurumi-ku, Yokohama, Kanagawa 230-0045, Japan
3) Graduate School of Frontier Sciences, The University of Tokyo, 5-1-5 Kashiwanoha, Kashiwa, Chiba 277-8561, Japan
4) Cooperative Major in Advanced Health Science, Graduate School of Advanced Science and Engineering, Waseda University, Tokyo, Japan
5) Broad Institute of MIT and Harvard, Cambridge, MA 02142, USA
6) Center for Computational and Integrative Biology, Massachusetts General Hospital and Harvard Medical School, Boston, MA 02114, USA
7) Department of Immunology, Weizmann Institute of Science, Rehovot 76100, Israel
8) RIKEN Center for Sustainable Resource Science, 1-7-22 Suehiro-cho, Tsurumi-ku, Yokohama, Kanagawa 230-0045, Japan
9) Department of Microbiology and Immunology, Faculty of Pharmacy, Mansoura University, Mansoura 35516, Egypt
10) Department of Gastroenterology, Osaka City University Graduate School of Medicine, Osaka, Japan
11) The Singapore Centre for Environmental Life Sciences Engineering, The School of Biological Sciences, Nanyang Technological University, Singapore
12) Microbial Genomics Section, National Human Genome Research Institute, National Institutes of Health, Bethesda, MD 20892, USA
13) Richard King Mellon Foundation Institute for Pediatric Research, Department of Pediatrics, University of Pittsburgh School of Medicine, Pittsburgh, PA 15224, USA
14) Graduate School of Environmental and Life Science, Okayama University, Okayama, Japan
*Corresponding authors’ email: [email protected]; [email protected]
This research was partly funded by Japan Agency for Medical Research and Development’s Core Research for Evolutionary Medical Science and Technology (CREST) and the Leading Advanced Projects for Medical Innovation (LEAP) programs.













