Aggregation of organic molecules shows strong absorption of near infrared light
Wed, Jun 14, 2017-
Tags
Possible developments in photovoltaic cells, sensors, and medical technology
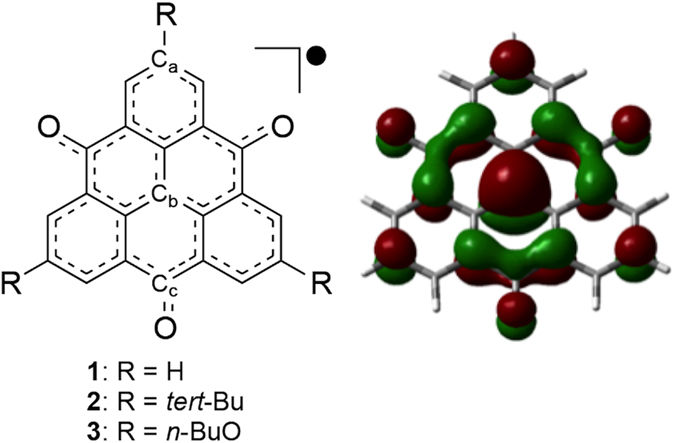
Chemical structure and SOMO of TOT. R denotes the substituent groups around the TOT skeleton.
A research team led by Professor Hiromi Nakai from Waseda University and Professor Yasushi Morita from Aichi Institute of Technology discovered that crystal structures of trioxotriangulene, an aggregation of organic molecules, can have strong near infrared absorption. In addition, the group revealed the mechanism of light absorption with quantum chemistry calculations based on originally derived methods.
This research was published in npj Quantum Materials on June 8.
Near infrared light, which has longer wavelength than visible light, is used in sensors and devices for optical communication. As potential material for such electronic devices, organic molecules that absorb near infrared light have been noticed. Materials made from organic molecules are light weight and flexible in use, but they can only absorb radiation with short wavelengths, such as ultraviolet rays and visible light. To stretch absorption wavelength to the near infrared region, it was necessary to enlarge molecules or add special substituents. However, these methods made synthesizing molecules and regulating the production and function of the devices difficult.
In this study, the group designed and synthesized two types of derivatives with substituents to trioxotriangulene (TOT), and they discovered that the crystal structures of these derivatives exhibit strong near infrared absorption. Furthermore, Professor Nakai conducted quantum chemistry calculations on the stacking columns extracted from these crystal structures and showed that showed that near infrared absorption occurs when electrons of TOT interact between protons.
Near infrared absorption of organic molecules realized in this study is based on a new mechanism that originates from an odd number of electrons of neutral radicals. The results of this research could be applied to develop photovoltaic cells and sensors using near infrared light and medical technology. Furthermore, there are great expectations for designing and producing organic electronic materials using quantum chemistry calculations.
Reference
- Published in npj Quantum Materials
- Title: Near-infrared absorption of π-stacking columns composed of trioxotriangulene neutral radicals
- Authors: Yasuhiro Ikabata, Qi Wang, Takeshi Yoshikawa, Akira Ueda, Tsuyoshi Murata, Kazuki Kariyazono, Miki Moriguchi, Hiroshi Okamoto, Yasushi Morita & Hiromi Nakai
- DOI:10.1038/s41535-017-0033-8













