
Proteins work in a folded state
Proteins, which play critical roles in living organisms, are linear polymers consisting of a string of amino acids. In order to function, proteins need to be folded into their unique 3D structures. Because strings of amino acids are unstable after they have just been synthesized, certain proteins called “molecular chaperones” attach to the strings and assist them to be correctly folded.
Most molecular chaperones are heat shock proteins (Hsps) and are present in every cell of living organisms from bacteria to human beings. Hsps are classified by molecular weight such as Hsp60, Hsp70, and Hsp90. Expression of Hsps increases when cells are exposed to some stress such as heat. Under such stress, more Hsps are generated to protect the cell because a cell cannot properly function when it has accumulated proteins that have degenerated due to stress. Such Hsps attach to degenerated proteins and restructure them into the correct structure to protect the cell. However, in cancer cells, Hsp70 helps the development and/or malignant transformation of the cancer.
This difference in function of Hsp70 is thought to arise from the difference in its interaction partners. We thus hypothesized that identifying and analyzing each different interaction partner of Hsp70 under various stresses would help elucidate the mechanism of stress response of cancer cells at the protein level.
Stress response factors that help the survival of colon cancer cells
Hsp70 has a variety of types that have similar structures and/or functions. We used one of them, Hsc70, in our experiment.
In the experiment, we subjected cancer cells to two types of stresses: serum starvation (no nutrients provided to cells) and anti-cancer drugs. We analyzed the detected Hsc70 complexes in these two experiments and found that under serum starvation stress, many Hsc70 combined with proteins that are involved in protein transportation in cells; under anti-cancer drug stress, many Hsc70 combined with proteins that are involved in metabolism. As we expected, Hsc70 combines with different proteins under different stresses.
Next, we explored the functions of proteins that combined with Hsc70 and found that Rab1A played an important role in stress responses among the 10 proteins that were commonly found to combine with Hsc70 under the two different stresses (starvation and anti-cancer drug). Initially, we subjected cancer cells to stress while suppressing the expression of Hsc70 and found degradation of Rab1A and death of the cancer cells. The fact that suppressing Rab1A expression causes cancer cell death indicates that Rab1A is an essential molecule for the survival of cancer cells and that Hsc70 helps the survival of cancer cells by suppressing degradation of Rab1A.
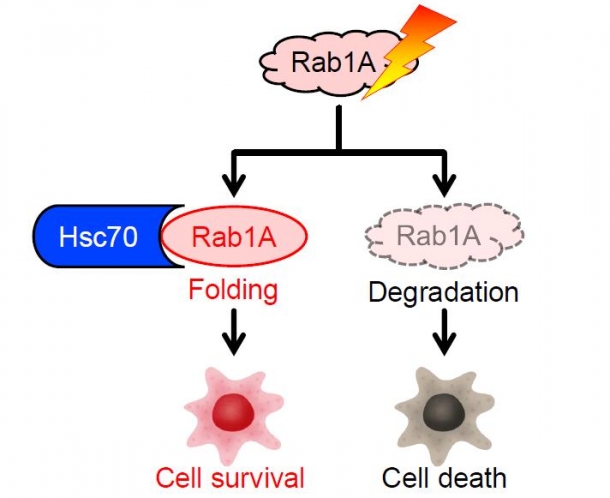
Fig. 1 Responses to stresses by Hsc70 and Rab1A
Hsc70 stabilizes Rab1A and hence confers stress resistance. Without Hsc70, Rab1A is degraded and cancer cells die.
Cancer drug resistance of scirrhous gastric cancers
In cancer treatment, cancer drug resistance, which means that the anti-cancer drug has diminished efficacy after repeated dosing, is broadly known. This resistance is also a phenomenon whereby cells develop resistance to stress, anti-cancer drug stress in this case, so we hypothesized that analyzing the client protein that combines with Hsp70 might lead to clarification of the mechanism of resistance.
We chose scirrhous gastric cancer, a type of cancer known to be difficult to treat, as the subject for the experiment. This type of cancer must often be treated with chemotherapy because it has already become impossible to treat surgically by the time it is found. In recent years, oxaliplatin has been approved for treating this type of unresectable advanced and recurrent gastric cancer. However, cancer drug resistance has been a major challenge in treating this type of gastric cancer. Cancer cells that have developed resistance can survive under doses of oxaliplatin at a higher rate than the original cells because such resistant cells have lost their sensitivity to oxaliplatin. We found that the SDF-2 gene is a novel resistant factor for cancer drug resistance by identifying multiple proteins that combine with Hsp70 in such resistant scirrhous gastric cancer cells and analyzing the roles they play.
Furthermore, we found that by suppressing the activity of the SDF-2 gene in resistant scirrhous gastric cancer cells so that SDF-2 proteins were not generated, the cells’ sensitivity to oxaliplatin increased and therefore their survival rate decreased. Even if scirrhous gastric cancer cells once develop oxaliplatin resistance, oxaliplatin recovers its efficacy for such cancer cells when SDF-2 is not active and hence the cells’ sensitivity to oxaliplatin is restored.
By comparing drug-resistant cancer cells and sensitive (non-resistant) cells, we found that resistant cells have a higher level of SDF-2 proteins (namely, such proteins exist in a more stable manner). This suggests that when scirrhous gastric cancer cells receive stress due to oxaliplatin dosing, Hsp70 combines with and stabilizes SDF-2 to confer drug resistance. This mechanism suggests that a novel cancer therapy that can control oxaliplatin resistance can be developed by setting SDF-2 proteins as a drug target.
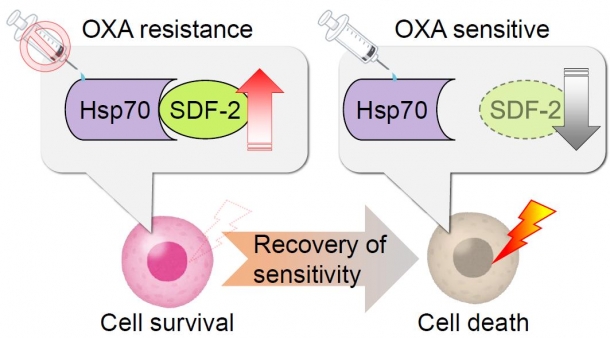
Fig. 2 Oxaliplatin resistance provided by Hsp70
Scirrhous gastric cancer cells develop resistance to oxaliplatin when Hsp70 combines with and stabilizes SDF-2.
Our analysis method, which utilizes the interaction between proteins and Hsp70, is innovative and different from conventional methods which utilize protein synthesis and gene expression, because it can comprehensively identify different proteins that are involved in different stress responses. Our method can be applied not only to cancer cells but also to diverse cells and thus is expected to contribute to the development of therapies for other diseases and understanding of biological phenomena. We intend to continue developing this method in the future.
Interviewed and written by Narumi Sato
In cooperation with J-School of Graduate School of Political Science, Waseda University









