A Three-dimensional Structure as a Previleged Structure in Medicinal Chemistry
Aromatic molecules include benzene, in which carbon atoms are connected in a hexagon arrangement, and compounds in which a functional group (a group of atoms) is attached to the apex of a benzene hexagon. Aromatic molecules are characterized by their planar (two-dimensional) structure. When a functional group is attached to one of these two-dimensional structure compounds from above or below the plane, a compound (alicyclic molecule) with a bumpy three-dimensional structure is formed. I am currently working to develop a method of converting two-dimensional aromatic compounds into three-dimensional compounds, a process called “dearomative functionalization” (Fig. 1).

Figure 1. Dearomative Functionalization
The molecule shown on the left in Fig. 1 is a benzene molecule, a representative aromatic molecule with a two-dimensional structure. A functional group, represented here by a colored ball, is attached to a two-dimensional structure to form a three-dimensional structure. Cyclic cmpounds but are not aromatic compounds are called alicyclic molecules.
In the drug discovery field, it is said that the overwhelming majority of excellent drug candidates are compounds with a three-dimensional structure. This is because the proteins in our bodies have a three-dimensional structure, so drugs with a three-dimensional structure work well since they tightly bind with proteins, like a key and a keyhole. In contrast, as a compound with a two-dimensional structure binds to various proteins like a master key, it sometimes causes side effects. However, because it is difficult to synthesize a compound with a three-dimensional structure, in the current situation, drug candidates are often compounds with a two-dimensional structure those are easy to synthesize. Although it is also possible to synthesize a drug molecules from a natural occurring molecules with a three-dimensional structure, there remains several problems such as the requirement of a large number of synthetic steps and the limitation of natural resources. For example, the flu drug Tamiflu (a three-dimensional structure) is made from natural shikimic acid, but it takes more than 10 steps to create it.
Attaching Functional Groups in the Intended Direction
At first glance, it seems very easy to break a two-dimensional structure to create a three-dimensional structure. However, since aromatic compounds are very stable compounds, it is very difficult to break their structures. Various methods have been tried, but there are problems: in particular, these methods require many highly toxic metal reagents; they require a large amount of starting materials; and they are conducted at high temperatures.
Therefore, in our laboratory, we have been conducting research towards efficient conversion of two-dimensional compounds into three-dimensional compounds using palladium metal as a catalyst. Recently, we have succeeded in developing a reaction called “dearomative alkylation” that attaches two functional groups to an aromatic compound, bromobenzene (Fig. 2).
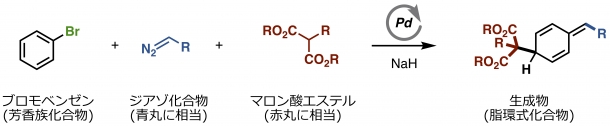
Figure 2. The Successful Dearomative Alkylation Reaction
From left to right:
– Bromobenzene (aromatic compound)
– Diazo compound (corresponding the blue ball in Fig. 1)
– Malonic acid ester (corresponding the red ball in Fig. 1)
– Product (alicyclic compounds)
Bromobenzene corresponds to the left object (aromatic compound) in Fig. 1, and the reaction product corresponds to the right object (alicyclic compound). Pd is the palladium catalyst. NaH is a substance required for this reaction.
Specifically, we found that the use of palladium as a catalyst can induce dearomatization (breaking a stable structure) of a planar molecule (Fig. 1, on the left), and can attach a functional group (Fig.1, red and blue balls) to the compound at the same time, synthesizing an alicyclic compound with a three-dimensional structure. So far, we have developed a proto-type reaction by using a compound other than malonic acid ester as the reactant corresponding to the red balls. However, this method has a problem in that it is difficult to convert the product into a more useful molecule. That is, after the dearomative reaction that attaches the red and blue balls shown in Fig. 1, when we performed an “derivatization reaction” to impart functional groups (represented by green and orange balls), we could not control the direction (pointing up or down with respect to the plane) in which each functional group attached. This causes various forms of 3D compounds to be created.
In that dearomative alkylation, there is a functional group called “ester” in the part corresponding to the red ball. Taking advantage of this ability to determine the direction in which other functional groups such as green and orange balls are attached during the derivatization reaction, we succeeded in creating a compound with a single type of three-dimensional structure. In other words, if we continuously perform our developed dearomative alkylation and the subsequent derivatization reaction, then by determining the direction of the functional group we can synthesize a structurally complex alicyclic compound with a high control of the direction of these functional groups (Fig. 3).
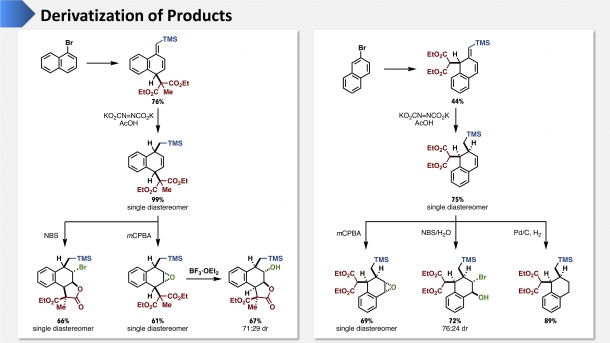
Figure 3. Various Compounds That Can Be Made from the Products of Dearomatic Alkylation Reactions
A two-hexagon substance with Br attached (a planar aromatic compound) serves as the starting material. The substances in the upper part of both the left and right figures of Fig. 3 are compounds formed by a dearomative alkylation reaction. Then by adding functional groups by means of various derivatization reactions, we create alicyclic compounds with complicated three-dimensional structures, as shown at the bottom of Fig. 3—and since the dotted lines are attached downward from the hexagonal plane and solid lines are attached upwards, clearly the molecule will have a bumpy structure.
Contributing to the Creation of a Library of Drug Candidates
In the future, I would like to make this reaction more general. In the currently successful dearomative alkylation, there is only one type of diazo compound for the blue ball and one type of malonic acid ester for the red ball. To overcome that limitation, we are conducting research to increase the number of types of compounds that correspond to the blue and red balls. We also intend to explore the use of catalyst metals other than palladium.
If this reaction can be generalized, it will be possible to create compounds with various three-dimensional structures. Pharmaceutical companies have many types of compounds in their libraries, and when they are developing a new drug, they begin by looking in the library for a compound that can bind to the protein of interest. We hope that our technology will enable the synthesis of many new compounds with three-dimensional structures for inclusion in those libraries. Drugs with new efficacy may be born from compounds with three-dimensional structures that were previously unimaginable.
Interview and composition: AIMONO, Keiko
In cooperation with: Waseda University Graduate School of Political Science J-School











