
Evolving cancer treatment
Professor Tasuku Honjo, who won the Nobel Prize in Physiology or Medicine 2018, discovered that protein PD-1 operates as a brake on the immune cells attacking cancer cells, and developed the discovery into an innovative cancer immunotherapy. Today, immunotherapy has become the fourth major treatment following surgery, radiation therapy, and chemotherapy.
While novel treatment methods have appeared, advanced cancers are still difficult to treat due to metastasis and/or drug resistance. In particular, pancreatic cancers are hard to treat even if they are found at an early stage. To overcome these cancers, I believe it is important that cancers do not develop in the first place. I thus started to explore the prophylactic treatment of cancers.
Mechanisms to suppress the development of cancers inherent in our bodies
Mutations in the genes of our bodies occur every day, resulting in mutant cells. However, mutant cells rarely grow into cancerous cells because our bodies have a mechanism to eliminate mutant cells. A typical example is attacks on mutant cells by immune cells.
Regarding eliminating mutant cells, it has been known for more than 20 years that normal epithelial cells push out mutant cells (Fig. 1). This is called the “cell competition” phenomenon, in which cells with different properties compete for survival in a limited habitat and the fittest (in this case, normal epithelial cells) survive. In cases where this mechanism does not work well, cancer develops, but the detailed mechanism was not understood. I, therefore, decided to elucidate the mechanism of cell competition in order to develop prophylactic cancer treatments.
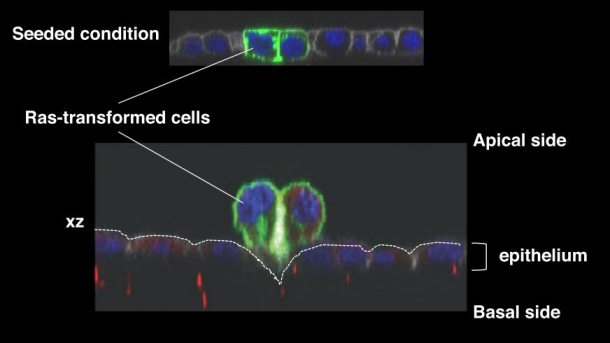
Fig. 1 Ras-transformed cells that are just being eliminated by normal epithelial cells
Elucidating cell competition phenomenon
Many cancers result from the accumulation of multiple gene mutations. Ras is known to be one of the genes that first mutate among such gene mutations. When a mutation occurs in the Ras gene, the relevant cell is not cancerous but is eliminated by normal epithelial cells. I explored the changes that occur in these normal and mutant cells at the time when a mutation occurs in the Ras gene.
I found that myosin accumulates in the Ras mutant cells specifically in the area that contacts with normal cells. This accumulation of myosin provides a slightly harder cell membrane compared to the other parts (Fig. 2). On the other hand, filamin accumulates in the surface area of the normal cells that contact with the mutant cells. It has been reported that these filamins provide the force to eliminate mutant cells physically. It has also been known that the accumulation of myosin in mutant cells triggers the accumulation of filamin in normal cells. Accordingly, I explored the substance that promotes myosin accumulation and successfully identified it. I then administered the substance to epithelial cells including mutant cells and found that the elimination of mutant cells was promoted as I had expected. This substance’s effect has been confirmed in mice, suggesting that the substance may be a candidate cancer drug, but the high toxicity of this substance remains a challenge.
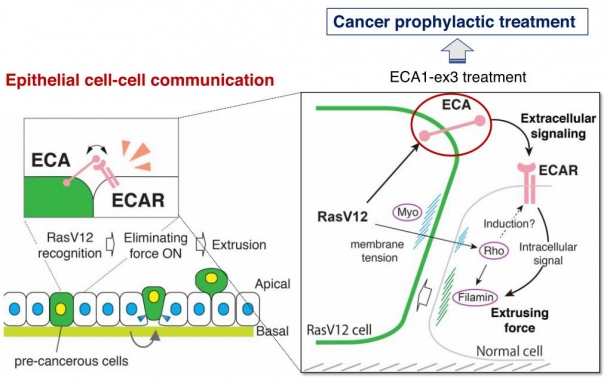
Fig. 2 ECAs and ECA receptors newly discovered in the surface of epithelial cells
Further study has revealed that myosin does not accumulate in Ras-mutant epithelial cells when they are not surrounded by normal cells. This suggests that there is some form of communication between normal and mutant cells. I have long been fascinated by the beautiful mechanism whereby dendritic cells communicate with T and B cells by presenting antigens in the immune system. I thus hypothesized that a similar mechanism might be at work in the cell competition phenomenon in epithelial cells.
This hypothesis turned out to be correct: I found that there are epithelial antigen-associated proteins (ECAs) on the surface of mutant cells, which correspond to antigens in the immune system, while there are ECA receptors (ECARs) that recognize ECA on the surface of normal cells. Furthermore, when ECA extracellular domains (a part of ECA that connects to ECAR) were added, the accumulation of filamin was facilitated in normal cells. Based on these observations, I have concluded that ECA generated by mutant cells facilitates the filamin accumulation in normal cells via ECARs.
Applicability to a wide range of medical fields
As the mechanism of intercellular communication is unveiled, drug companies have started to express interest in prophylactic cancer treatment utilizing this mechanism. This makes my research exciting because I feel that I am getting close to the truth of intercellular communication. In the future, I would like to develop this research outcome into a cancer-preventive drug that people can take daily just like dietary supplements.
Intercellular communication is also thought to be related to many diseases other than cancers. For example, the mechanism of eliminating virus-infected cells is likely to involve communication between infected and uninfected cells. The phenomena whereby transplanted cells are pushed out by surrounding cells and fail to be engrafted, and neurons are destroyed in Alzheimer’s disease patients, may also involve intercellular communication. I hope that research on intercellular communication advances, leading to treatment methods for many diseases.
Interview and Composition:Akiko Ikeda
In cooperation with: Waseda University Graduate School of Political Science J-School









