
Diverse materials that support life
We are surrounded by many materials with interesting properties. There are conductive and nonconductive materials, and magnetic and nonmagnetic materials. Even the same material can display different properties when the temperature or pressure is changed (this is called a phase transition). Humans have utilized such materials to create useful products.
For example, a maglev (linear motor car) makes use of superconductive materials. Superconductors are materials that provide zero electrical resistance at extremely low temperatures. In the superconducting state, the material does not heat up when current is passed through it; thus, by using a large current, a strong magnet can be created. Through the use of superconductors, 500 km/h high-speed transportation is becoming more tangible.
How are materials created? I simulated the behavior of electrons using a supercomputer to examine this principle. If we determine the principle behind creating materials, we can make predictions such as, “if we synthesize such a material, it will have such properties”.
Understanding a material from the behavior of electrons
Let us look at materials from a microscopic viewpoint. A material is an aggregate of atoms, and there are a large number of electrons around the atoms. Depending on the behavior of these electrons, the material’s properties change notably.
For example, conductivity is determined by the freedom of electrons to move. If you consider the image in Figure 1, this principle becomes easier to understand.
Imagine that electrons can sit on two-person couches (electron orbitals). If there are empty spots, the electrons can change spots (as in a metal: Figure 1, left), but if all the spots on the couches are filled, the electrons cannot move (as in an insulator: Figure 1, center). However, even if there is an empty spot on a couch, if the repulsion between the electrons on that couch is too strong, an electron cannot sit there; thus, it cannot move (Figure 1, right). Such a state is called a Mott insulator, and is the topic of my research.
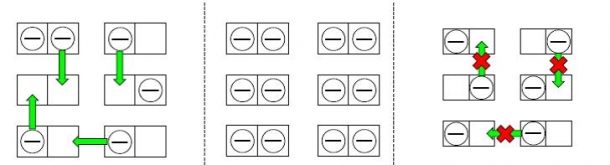
Figure 1: The left image shows electrons in a conductor. Since there are empty spots on the two-person couches, electrons can move freely. The center image shows electrons in an insulator. The couches are filled with electrons, meaning the electrons cannot move. The right image appears to show the potential for conductivity, but actually represents a Mott insulator. When the repulsion between the electrons is too strong, even if there are free spots on the couches, the electrons cannot sit next to each other, meaning they cannot move. The number and size of couches are determined individually for each material (Image source: Hiroshi Watanabe, assistant professor).
The property of a material being attracted to magnets (magnetism) can be explained by the behavior of electrons. Electrons spin, and because they are negatively charged, spinning makes them act as small magnets. Depending on the direction of spin, the electron may become an upward magnet (up spin) or downward magnet (down spin) (Figure 2, top). When the number of up spin and down spin electrons are the same, the material is not magnetic (Figure 2, center), but if spins are aligned in the same direction, it becomes magnetic (Figure 2, bottom).

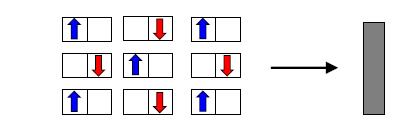
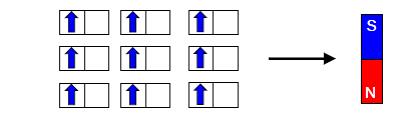
Figure 2: The relationship between magnetism and electron spin. In many cases, there are the same number of spins in each direction (center), in which case the material does not display magnetism. Under unique conditions, a couch is filled with electrons facing the same direction, making the material magnetic (bottom) (Image source: Hiroshi Watanabe, assistant professor).
The spin direction of electrons is determined by many elements interacting in a complex manner. I am studying how to express such behavior in terms of formulas.
Elucidating a unique insulating mechanism and predicting conductivity
I studied the compound Sr2IrO4. It was already known that this compound is an insulator, but there was not yet an explanation for why this was the case.
I focused on an iridium (Ir) atom, and the manner in which the 5d electron associated with magnetism sits on a couch (electron orbital). To calculate the state of the electron on the 5d orbital, I assumed that the effect of the “spin-orbit interaction (λ)” must be taken into account. The spin-orbit interaction indicates the spin direction affected by the electron orbital. Normally, λ is much smaller than the “Coulomb interaction (U)” between electrons; thus, it does not typically affect the calculation if ignored. However, on the 5d orbital, U is comparable in size to λ.
When λ was used in the calculation, we were able to explain the Sr2IrO4 compound being an insulator (Figure 3). When the spin-orbit interaction is included, the calculation becomes more complex than usual. However, we were able to use this study experience to derive a method of performing such a calculation effectively.
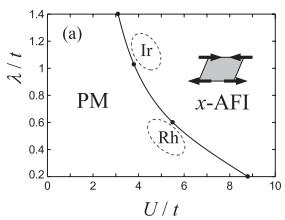
Figure 3: Impact of the spin-orbit interaction (λ) on the prediction of the insulating property of a material. PM is the metal region and x-AFI is the insulator region. t is the kinetic energy of the electrons, which is used as the unit for energy. When the effect of λ is incorporated (λ/t becomes large), the material becomes an insulator even if the Coulomb interaction (U/t) is small. Since Ir has a large spin-orbit interaction (λ/t is near 1–1.2), the material can be an insulator despite the Coulomb interaction not being very large (U/t is around 4–6). A system in which U is smaller than t is called a weakly interacting system, while one where ,U is larger than t is called a strongly interacting system. Sr2IrO4 is in the “intermediate interaction” range, where interactions change from weak to strong. (H.Watanabe, T. Shirakawa, and S. Yunoki, Phys. Rev. Lett. 105, 216410 (2010))
We noticed that the crystal structure and electron state of Sr2IrO4 are similar to those of La2CuO4, the original high-temperature superconductor. Thus, we wondered if, by changing the electron density, Sr2IrO4 might also become a high-temperature superconductor. We therefore applied the computer simulation that I developed. At an electron density of five, Sr2IrO4 is an insulator, but when the electron density is increased by about 0.2, it may become a superconductor (Figure 4).
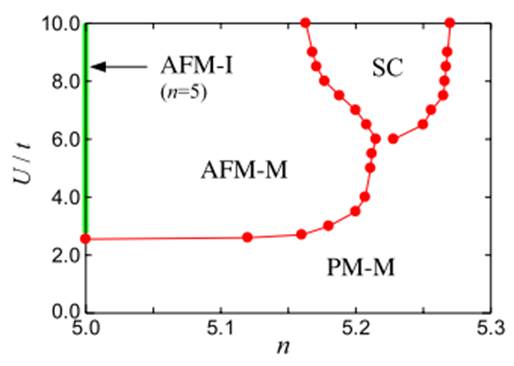
Figure 4: The relationship between the behavior of Sr2IrO4 and the electron density of the material, where n is the electron density and U/t is the Coulomb interaction. PM-M is the paramagnetic metal region, AFM-I is the antiferromagnetic insulator region, AFM-M is the antiferromagnetic metal region, and SC is the superconducting region. When synthesizing a compound, if some of the strontium and iridium can be substituted by an atom with many electrons, the electron density (n) can be increased. The Coulomb interaction (U/t) can be adjusted by both the pressure and replacement of atoms. (H.Watanabe, T. Shirakawa, and S. Yunoki, Phys. Rev. Lett. 110, 027002 (2013))
Dreaming of an unimaginable material
Experimental physicists are working every day to synthesize the superconducting material that I have envisioned. However, increasing the electron density turns out to be difficult in practice, and such a material has not yet been synthesized. If it is actually synthesized, we can confirm its properties. I am patiently waiting for that day.
Theoretical physicists such as myself can calculate the properties of hypothetical materials by inputting electron density using a keyboard. Thinking about the number and state of electrons that could produce this interesting material is the essence of my study, which is why it is so enjoyable. We may be able to discover unknown properties and phase transitions beyond ferromagnetism and high-temperature superconductivity. If we discover such a material, we may be able to change the world. For example, there may be a material that could be used to build a computer with much higher performance and speed than exists today. We may be able to create a material for power lines that is able to transport electricity over long distances without any loss. I will continue with my work with faith that such a material will one day be developed.
Interview and Composition:Kaori Oishi
In cooperation with: Waseda University Graduate School of Political Science J-School










